Antimony

When used as an alloying element, antimony greatly increases the hardness and mechanical strength of lead. The main use of antimony is to harden lead in storage batteries, and it is increasingly being used in the semiconductor industry.
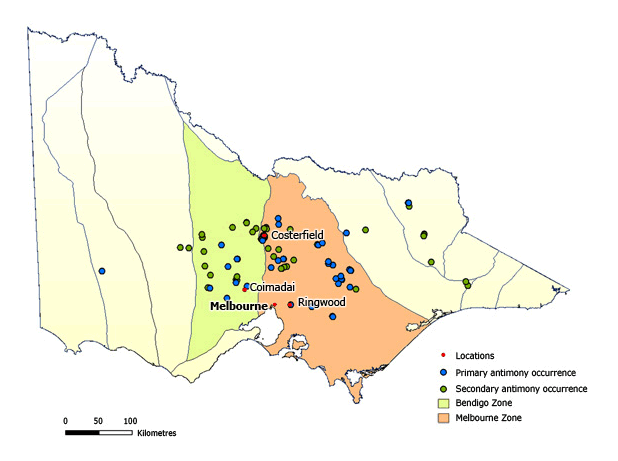
Historically and economically, antimony is the second most important metallic commodity in Victoria, after gold. The only significant ore mineral of antimony, stibnite (Sb2S3), is widespread as an accessory mineral in gold-bearing quartz veins, particularly in veins that intersect the Silurian – Early Devonian Murrindindi Supergroup sedimentary rocks of the Melbourne Zone.
Deposits are mostly in the form of reefs, shear lodes and fissure fillings, close to the hinge zones of folds within sedimentary rocks, or associated with dykes. The sediment-hosted antimony deposits are epithermal, formed from hydrothermal solutions related to deformation of the Palaeozoic rocks.
A significant antimony resource is located at Costerfield. In addition, there has been important historical production from the Ringwood and Coimadai deposits.
Stibnite is also a minor constituent of gold-bearing sulphide reefs of the Bendigo Zone and there is bournonite (PbCuSbS3) in the silver–lead ores of eastern Victoria.
Further information
Minerals of Victoria - Geological Survey of Victoria Report 92
Maps
To create your own maps online and in real time, plan exploration activities by viewing land status, or download GIS data to add to your own maps, visit GeoVic.
Page last updated: 12 Jan 2023